Chemistry - Molecular Structure (35 of 45) s-p3-d2 Hybridization - Sulfur Hexafloride - SF6 - YouTube

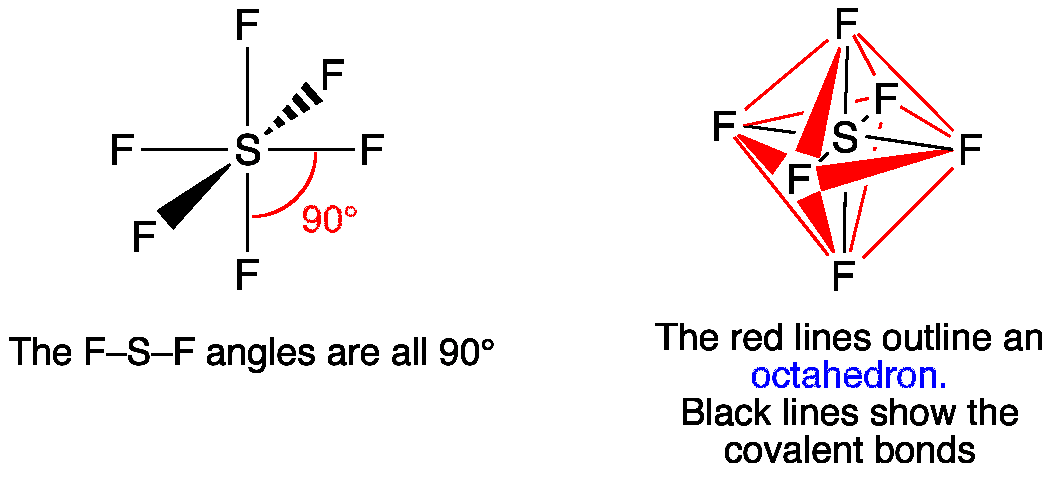
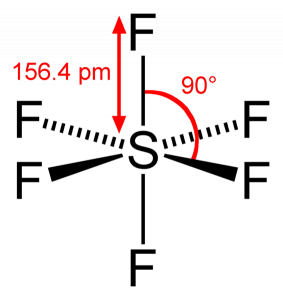
Describe hybridisation in the case of PCl5 and SF6. The axial bonds are longer as compared to equatorial bonds in PCl5 whereas in SF6 axial bonds and equatorial bonds have the same

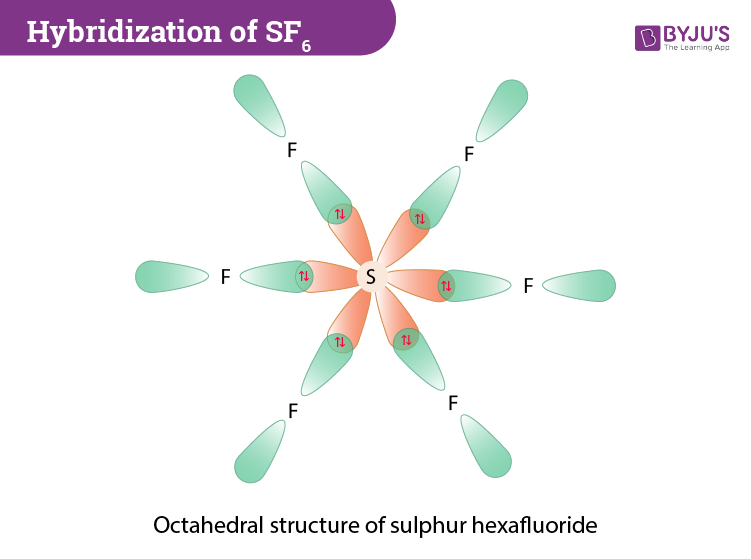
The sp3d2 Hybridization and Octahedral Geometry | Molecular geometry, Chemistry, Electron configuration
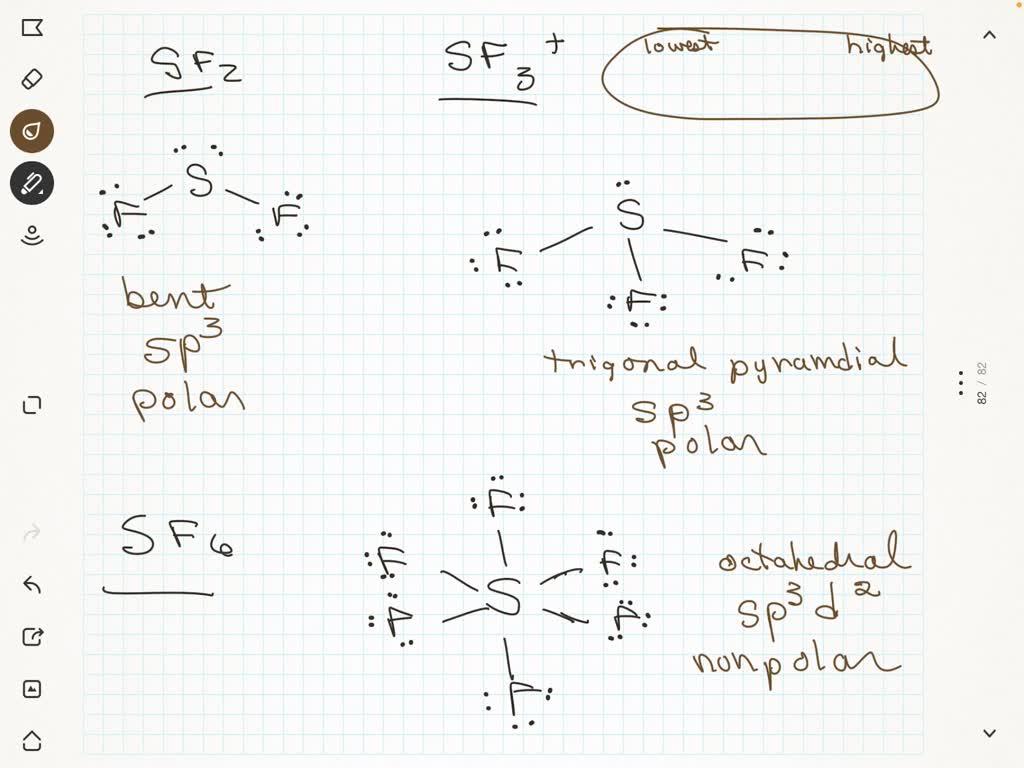
SOLVED: Consider the following molecules: SF2, SF3+, SF6. Draw the Lewis dot structures, determine VSEPR shapes, give hybridization of the central atoms, and predict whether the molecules are polar. Put in order

SOLVED: What is the hybridization of the central atom in SOF4? Hybridization What are the approximate bond angles in this substance? Bond angles What is the hybridization of the central atom in
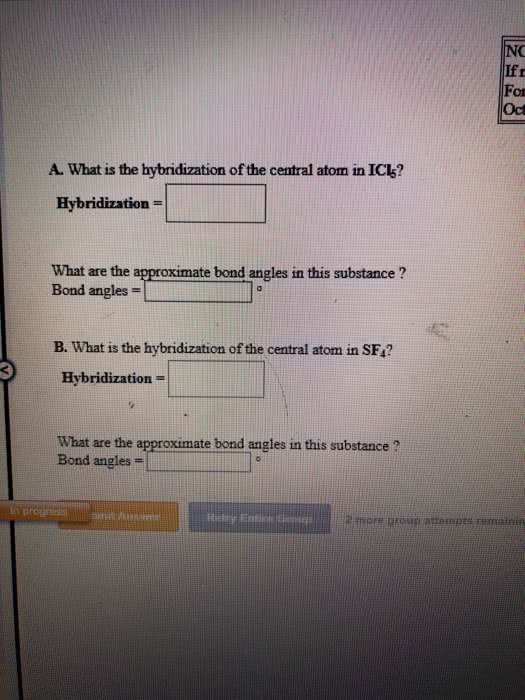
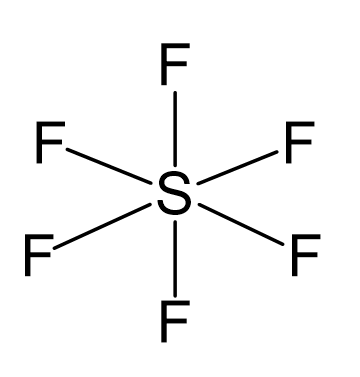
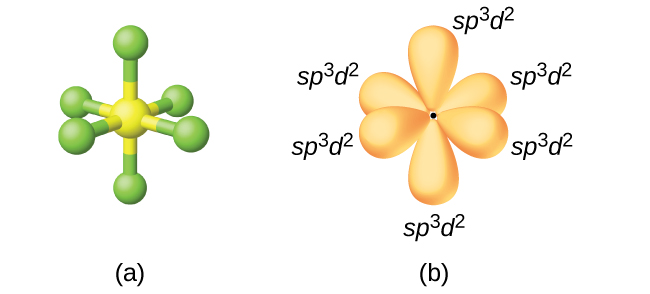
OneClass: NOTE If more For exa Octahe A. What is the hybridization of the central atom in SF6? Hybrid...

SOLVED: 2. Give the expected hybridization of the central atom for the molecules below: a. SF6 b. ClF5 c. XeF4

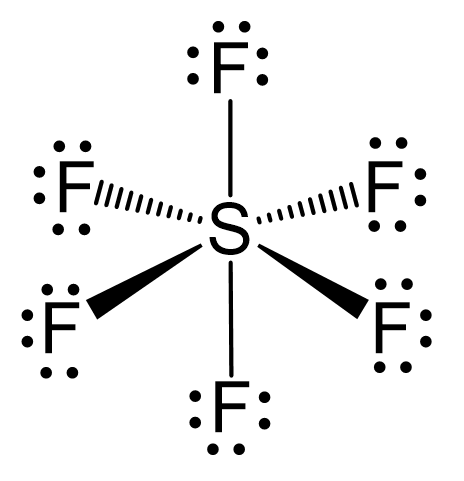
What atomic or hybrid orbitals make up the sigma bond between S and F in sulfur hexafluoride, SF6? | Homework.Study.com